Corrosion Is A Stealthy Hazard Dont Ignore It In Your Laboratory
The proverb “a stitch in time saves nine” is an apt reminder for the laboratory users. You can prevent many incidents in your laboratory through proactive actions. Watching out for corrosion in the laboratory should be high on your safety agenda.
LABORATORY HAZARDS
A laboratory, compared to an average workplace, is teeming with risks and hazards. Every aspect of the laboratory – infrastructure, equipment, consumables, operation procedures & process, and lab users – is risk prone. The slightest mistake may lead to hazardous accidents.
Knowing the various hazards helps understand take preventive steps to avert an incident.
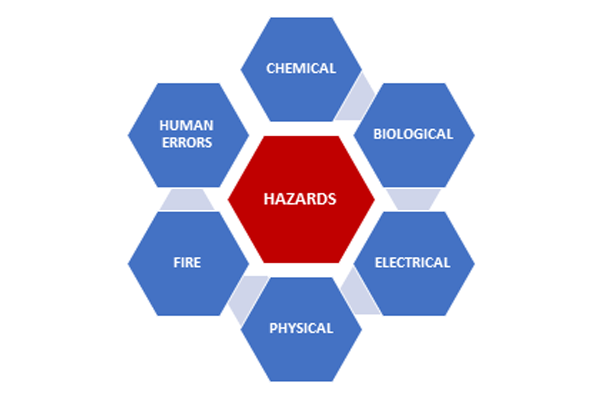
CHEMICAL HAZARD
Chemicals use is an essential part of the research process. Many chemicals (solid/liquid & gases) used in the labs are corrosive/toxic and can pose a severe health risk if improperly handled. From storing the chemicals to using them in the experiments and disposing them, extreme care and safety precautions will have to be followed.
BIOLOGICAL HAZARD
Laboratories handling biological agents – such as blood, body fluids, pathogens, experimental animals – are prone to biological hazards. Scientists researching and working with bio agents are exposed to the threat of bacterial and viral infections, allergens, and microbes. Like chemicals, biological safety requires careful consideration from samples collection till safe disposal.
ELECTRICAL HAZARD
Electricity powers laboratories and overlooking electrical safety can prove deadly. Shock and electrical fire are the common hazards encountered in the laboratory. Care should be taken whilst using electrical appliances – dangling and intrusive electrical cords, malfunctioning appliance, exposed or broken points, and liquids near electrical units – to prevent accidents.
PHYSICAL HAZARD
Laboratories are buzzing with activity. It’s unimaginable the number of minor mishaps leading to a severe risk to human and non-human resources. Wet floors, heat burns, electrical shocks, chemicals burn, broken apparatus, radiation, injury from machines with moving parts, and overexposure to noise are common physical threats in a laboratory.
FIRE HAZARD:
Fires can devastate the safety and operations of a lab. Explosion from chemical reactions, short-circuit and untended electric lines and equipment, flammable liquids and solvents are the common factors leading to fire. Fire poses a severe threat to human life and can cause extensive damages to the lab infrastructure.
HUMAN ERRORS:
Human oversight is a leading factor for laboratory safety incidents. Despite the advances in technological solutions to minimise human factors, safety incidents continue to happen. Improper usage of equipment, a momentary lack of concentration, slippages in adhering to safety protocol are key contributors for human errors.
CORROSION – THE STEALTHY HAZARD IN YOUR LAB
Having known the various laboratory hazard, let’s now turn our focus to corrosion.
Some hazards are momentary – they happen in-the-moment. While others lurk in the background, waiting to explode one day. Corrosion lurks and builds up. The resulting damage from corrosion can be devastating.
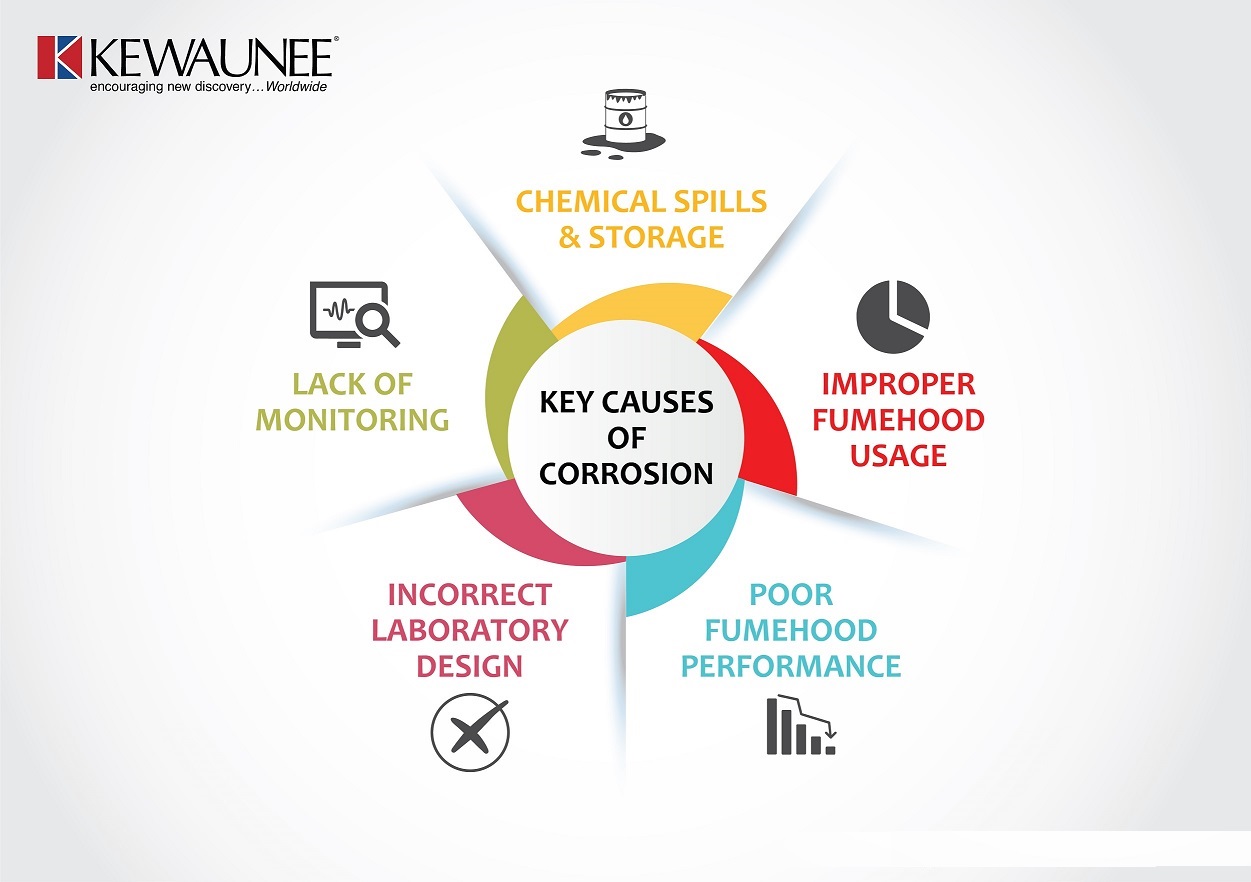
Myriad factors contribute to corrosion. Let’s examine the key ones.
1. CORROSION PRONE AREAS CHECK:
The common lab areas prone to corrosion are fume hoods, sink area, and the chemical storage cabinets. Lab users should exercise care to follow all operational guidelines of the equipment and safety protocol of the lab areas.
Turning on the exhaust, while washing at the sink, is a simple yet effective procedure to prevent the sink area corrosion. The subsequent points cover the chemical storage, fume hood and others in detail.
2. CHEMICAL SPILLS & HOUSEKEEPING:
Laboratories handle chemicals in their day-to-day research and experiments. Many chemicals are corrosive, and spillages can prove detrimental to the structural integrity of the lab furniture and equipment.
Choice of right material of construction, based on the lab application, is critical during the lab planning and design phase. In addition, provisions to treat any exposure or clean spillages should be part of laboratory housekeeping standard operating procedure (SOP).
Lab users, scientists, and facility management personnel must be trained in all safety procedures and made aware of the harmful effects of corrosion.
3. CHEMICAL STORAGE:
Storing chemicals in the laboratory needs special attention. Care should be exercised to check for appropriate storage facility (flammables storage and acid/corrosive storage cabinets).
Using spill-proof, non-breakable, and secondary containers to catch spills will prevent corrosion in the storage areas. Sealing the containers to prevent vapours escape and not using fumehood as a storage facility can enhance the longevity of lab equipment.
4. IMPROPER FUME HOOD USAGE:
Fume hood (fume cupboard or fume closet) is a local ventilation device found in all laboratories. Ventilation devices are used to capture and remove the contaminants (gas, vapour, or dust) away from the users and out of the laboratory. While operating the fume hoods, lab users must adhere to the best practices.
Keeping the fume hood sash open for longer than necessary or continuously above the safe height may lead to inefficient containment. Long exposure to residual fumes leads to corrosion in the exposed areas.
Similarly, switching off the fume hood when the laboratory is not in use, may lead to acid rain phenomenon in the lab. The unexhausted chemical residue hanging in the unventilated lab will lead to corrosion. It’s a good and a recommended practice to keep the fume hoods on even during lean hours. Using fume hood nighttime setback option preserves some flow through the empty lab while saving energy.
5. POOR FUME HOOD PERFORMANCE:
Fume hoods operate at a defined range of face velocity 80 FPM to 100 FPM (measured in feet per min or mtrs/sec) for effective containment. Besides improper usage, if fume hoods are not adequately maintained or handled poorly, it may lead to drop in fume hood efficiency. An inefficient fume hood results in sub-par containment. When the contaminated fumes are not exhausted well, over a long period, the fumes will end up corroding the exposed laboratory infrastructure.
Choosing the right fume hood, lined with non-corrosive materials, is also critical in enhancing the fume hood performance. ASHRAE 110 -2016 & EN 14175 provides detailed guidelines on how to test fumehoods for the best containment.
6. INCORRECT LABORATORY DESIGN:
Laboratories, specifically chemical labs, are designed to operate as negative pressure zones. In a well calibrated and controlled negative pressure zone, the air flow prevents harmful cross-contamination. In a negative pressure zone, airflow will be from low-hazard to high-hazard areas removing the corrosive chemicals out of harm’s way.
Factors such as laboratory space, equipment, nature of experiment, and usage impact the contamination requirements of the laboratory. During the lab design and planning, extreme care must be exercised for calculating the negative pressure requirements in various lab usage scenario.
The critical elements to be considered during lab design are:
7. ENVIRONMENT MONITORING:
During operational phase it is critical to ensure a corrosive free lab. Continuous monitoring of the internal lab environment – such as air quality monitoring, volatile organic compounds (VOC) levels, optimal temperature etc – should be in place.
Adhering to the global and local safety standards and limits, alerting lab users in the event of safety violations, and preventing disasters should be a part of the operational culture.
CONCLUSION
In a volatile environment such as a laboratory, hazard can happen anytime. Some hazard indicators are visible, and some are not. Certain incidents happen immediately, and some takes months and years to build up to a disaster.
The only solution is always to remain vigilant and prevent incidents. Remember, hazard assessment is not putting up a safety manual at the start of the lab operations. But, a continuous process as part of the laboratory safety manual, we should make periodic and regular inspection for corrosion across the laboratory mandatory.
Comments are closed.











